
Photocatalytic Reduction of CO 2 into CH 4 Using SrB 2O 4 Catalyst GUO Li-Mei, KUANG Yuan-Jiang, YANG Xiao-Dan, YU Yan-Long, YAO Jiang-Hong, CAO Ya-An. Structures, Stabilities and Physicochemical Properties of Nucleobase Tautomers Second-Order Nonlinear Optical Properties of Bis-Cyclometalated Iridium(Ⅲ) Isocyanide Complexes ZHU Chang-Li, WANG Wen-Yong, TIAN Dong-Mei, WANG Jiao, QIU Yong-Qing. WANG Yu-Qiao, WANG Pan-Pan, LU Jing, BAI Yi-Chao, GU Yun-Liang, SUN Yue-Ming.ĭye-Sensitized Solar Cells Based on MWCNT/TiO 2 Counter Electrode and Thiolate/Disulfide Non-Iodine Redox Couple


Redox-Responsive Interfacial Properties of Se-Containing Sulfobetaine Surfactant Wei-Wei KONG,Shuang GUO,Yong-Min ZHANG,Xue-Feng LIU. Hui-Jun YAN,Biao LI,Ning JIANG,Ding-Guo XIA.įirst-Principles Study:the Structural Stability and Sulfur Anion Redox of Li 1- xNiO 2- yS y. Acta Physico-Chimica Sinica, 2019, 35(12): 1372-1381.Ĭhunhe YANG,Aiwei TANG,Feng TENG,Kejian JIANG.Įlectrochemistry of Perovskite CH 3NH 3PbI 3 Crystals Ultra-High Proton/Vanadium Selectivity of Polybenzimidazole Membrane by Incorporating Phosphotungstic Acid Functionalized Nanofibers for Vanadium Redox Flow Battery Xiaobing YANG,Lei ZHAO,Xulei SUI,Linghui MENG,Zhenbo WANG. Recent Advances in Polyoxometalates for Applications in Electrocatalytic Hydrogen Evolution Reaction Jingxuan Ge, Jun Hu, Yingting Zhu, Zonish Zeb, Dejin Zang, Zhaoxian Qin, Yichao Huang, Jiangwei Zhang, Yongge Wei. Redox-Regulated Dynamic Self-Assembly of a Lindqvist-Type Polyoxometalate Complex Jing Zhang, Lina Wang, Xiaofei Chen, Yufeng Wang, Chengyan Niu, Lixin Wu, Zhiyong Tang. Journal of Guangxi Normal University: Natural Science Edition, 2009, 27: 78 Journal of Hebei University: Natural Science Edition, 1997, 17: 73 Ģ. Key words: Hydrophobic amino acid, Ionization potential, Electron affinitiy, Redoxġ. The molecules of the hydrophobic amino acids are oxidized or reduced with difficulty in gas phase because of their high VIPs and negative EAs. The electron affinities (EA) for the amino acids are small and negative ranging from -0.08 to -0.63 eV. The attachment of one electron to the molecules of hydrophobic amino acids leads to anions in which the extra electron is bound to the H atoms of the carboxyl or amino groups and to their valence orbitals, reflecting the double nature of the dipole -bound state and the valence state. The AIPs of cysteine, methionine, phenylalanine, tyrosine, and tryptophan decrease because of the larger amount of negative charge removed from the atoms in their side chains. This yields large adiabatic ionization potentials (AIP) of 8.52-9.15 eV. For glycine, alanine, proline, valine, leucine, and isoleucine with small side chains, the computational results indicate that the negative charges are removed from the atoms of their amino, α-carbon, and carboxy moieties in one electron oxidation reactions.
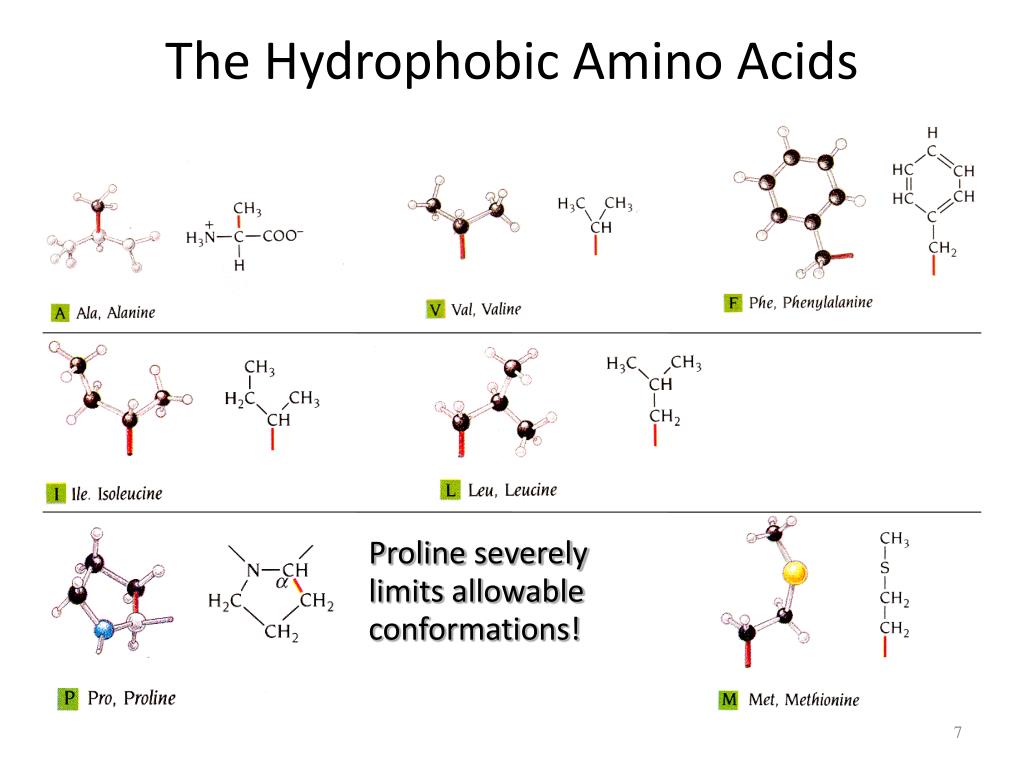
Characteristics of the one electron redox behavior of hydrophobic amino acids in gas phase were calculated with density functional theory at the B3LYP/DZP++ level.
